 His work during WWII led to the development of the atomic bomb, and his research on uranium and plutonium fueled the creation of nuclear power plants. He proposed the idea of a fusion reaction, which is when two nuclei are combined to form a larger nucleus. Niels Bohr also made great contributions in the production and utilization of nuclear energy. Production and Utilization of Nuclear Energy This meant that physicist had to incorporate considerations from both classical physics and quantum theory when studying physical phenomena, leading to greater progress in understanding the universe around us. These theories highlight the fact that physical systems such as atoms cannot be described using solely one type of description rather, a balance between both wave and particle descriptions must be considered. The Principle of Complementarity and Wave-Particle Duality Theory were two of the major contributions made by Niels Bohr. This knowledge played a vital role in our understanding of chemistry and allowed us to explain phenomena like spectral lines.Ĭomplementarity Principle & Wave-Particle Duality Theory. He also discovered the mathematical formula for calculating the energies of these electron shells and the frequencies at which electrons move between them. This theory led to the discovery of electrons being in various concentric shells around a nucleus, rather than in just one general area. He developed a deep understanding of how electrons behave in atoms and made the foundational theories that are still used today.īohr’s most groundbreaking and noteworthy contribution was a theory of atomic structure, an idea which he put forth in 1913. In 1913, he proposed his revolutionary atomic model which helped shape our current understanding of chemistry, physics, and even biology. Niels Bohr was a Danish physicist who made very important contributions to the understanding of quantum mechanics. Introduction to Niels Bohr’s Contributions in chemistry In 1913, he proposed his atomic model and earned the 1922 Nobel Prize in Physics for his work in developing quantum theory. Niels Bohr was a Danish physicist whose groundbreaking atomic models have revolutionized our understanding of the physical universe. In 1913, Niels Bohr developed his groundbreaking atomic model and revolutionized our understanding of the physical universe. Thus frequency and wavelength are inversely proportional to each other. That outermost shell(orbit) cannot accommodate more than 8 electrons.Ĭ=vλ where c= speed of light and λ is wavelength. Niels BohrĮlectrons are found by 2n 2 formula in different shells where n=shell Once they move from one orbit to another they emit energy.  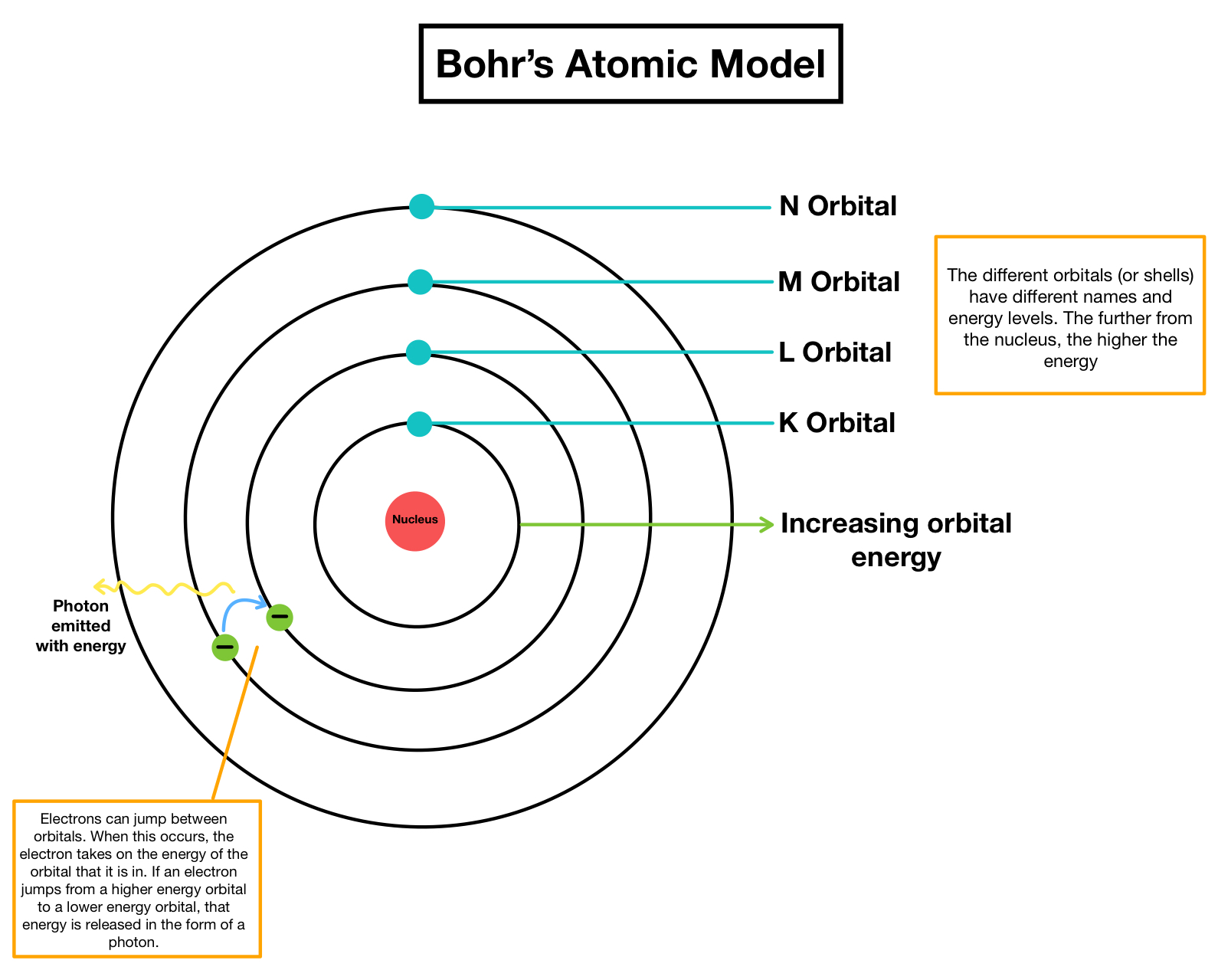
Niels Bohr Contributions to Quantum Mechanics improved Rutherford’s nuclear model by suggesting that the electrons revolve in the discrete energy orbits and they do not loose energy while they are in the orbit. TOK connections in stoichiometric relationships. 
PPT 10.1 Fundamentals of organic chemistry.PPT 8.4 Strong and weak acids and bases.PPT on 1.1 Particulate nature of matter.Bohr used his model to explain the spectral lines of hydrogen. This atomic model was the first to use quantum theory, in that the electrons were limited to specific orbits around the nucleus. This means that electrons’ energy levels are discrete and that the electrons revolve in stable orbits around the nucleus, but can jump from one energy level to another. As part of his contributions, Bohr developed the ‘Bohr Model’ of the atom, in which he proposed that the electron was able to occupy only certain orbits around the atomic nucleus. 
At the university, he came under the guidance of Prof C Christiansen, and earned his Master’s degree in Physics in 1909 and Doctorate in 1911. In 1903, Bohr enrolled as an undergraduate at the University of Copenhagen, majoring in physics, while also studying astronomy and mathematics. Bohr was born in Copenhagen on October 7, 1885, the second of three children of Christian Bohr, a professor of physiology at the University of Copenhagen, and his wife Ellen. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
